Advance your ocular therapies with the only full-service contract research organization to offer personalized ophthalmic drug development services that span the entire continuum from discovery and preclinical to clinical trials and post-market follow-up studies.
Whether you need support with anterior or posterior segments, rare ocular diseases, gene therapies or ophthalmic devices and diagnostics, we know how to optimize your ophthalmic development.
眼科プログラムのための戦略的ビジョン
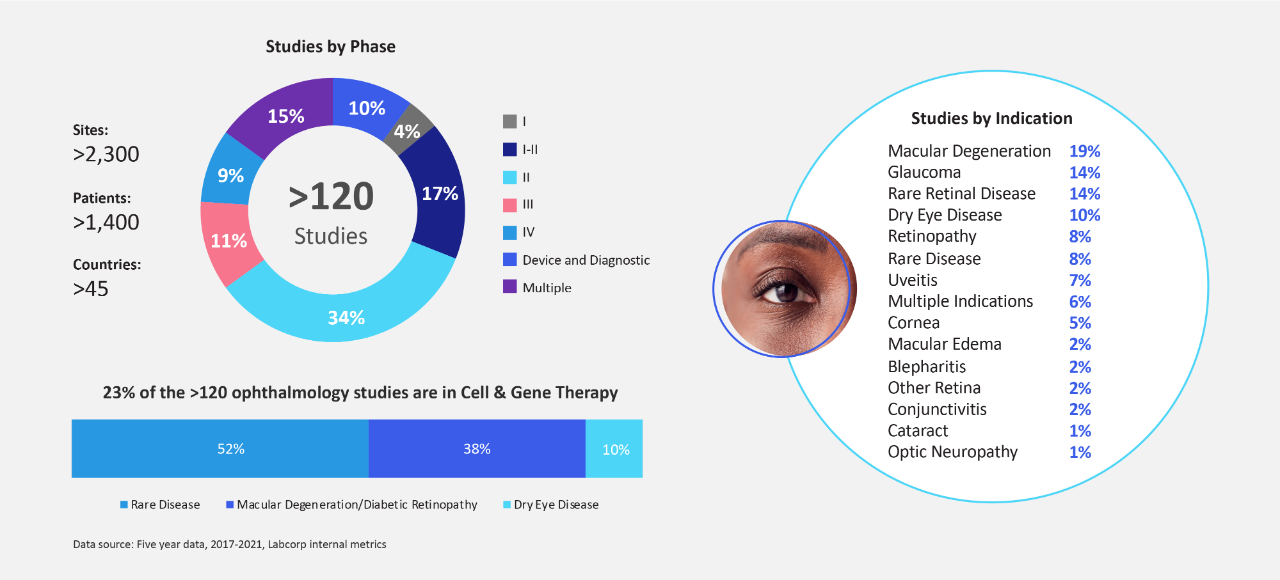
当社の眼科スタッフの科学および規制に関する深い知識と経験をご活用ください。当社スタッフはこの 5 年間で、新興バイオテクノロジー企業向けに患者様中心のグローバルな眼科治験 120 件以上に携わってきました。
Work with renowned vision scientists as part of our more than 30-year relationship with Ocular Services on Demand (OSOD) to handle your preclinical ocular needs and deliver specialized expertise in the ocular surface and anterior and posterior segments
Enable intelligent patient recruitment with advanced data and increase your efficiencies with our established relationships with more than 2,000 global sites
Extend your ophthalmic team’s expertise with a full-service collaboration
患者様のニーズを意思決定の最優先事項にして眼科医薬品開発プログラムを成功させようとするときには、経験が重要になってきます。ラボコープはこの 5 年間、新興および中規模のバイオテクノロジー企業に柔軟なソリューションを提供し、その専門知識と洞察で 120 件以上の眼科臨床研究に貢献してきました。
さまざまな部門にまたがるサポートとカスタマイズされた戦略によって、開発スケジュールの迅速化、問題の予測・防止を図り、眼科医薬品の市場投入を成功に導きます。お客様のチームが当社と強力なパートナーシップを結ぶことで、患者様の暮らしをより速やかにより良いものにすることができます。
お客様のニーズに合わせた前臨床ソリューション
You may need toxicology (good laboratory practices and non-good laboratory practices), tissue distribution/pharmacokinetics and pharmacology to support your ophthalmology preclinical research. 当社は、眼表面と前眼部、後眼部それぞれの違いを理解しており、緑内障、ドライアイ、ブドウ膜炎、アレルギー、網膜疾患など複数の眼疾患領域の専門知識を提供することができます。
また、当社にはすべての医薬品 / 治療クラスを扱った経験があり、OSOD とも業務提携しているため、数多くのモデルで眼球からの投与経路を提案することができ、前臨床のプロセスで役に立ちます。
ラボコープと一緒に眼科学的研究に取り組むことで、貴社のチームは当社のフルサービスのラボを利用して、化学分析サービスやバイオ分析サービスのあらゆるニーズに対応できるようになります。統合された包括的なサービスの違いをご体験ください。
Unique solutions for clinical trial testing
当社は、お客様のプログラムのニーズに柔軟に対応する、患者様中心の独自の治験および試験ソリューションの提供を第一に考えています。
- Gain scientific and regulatory support – Extend your team's capabilities with our regulatory and commercial guidance. Count on our extensive experience in assisting both small biotech and large pharmaceutical companies to help you achieve your strategic goals
- Apply flexible, integrated delivery models – Whether you need to apply decentralized clinical trial approaches to mitigate delivery risks, we can work together to optimize your delivery
当社では、お客様のご希望やプロジェクトのニーズに合わせてサービス内容を調整します。